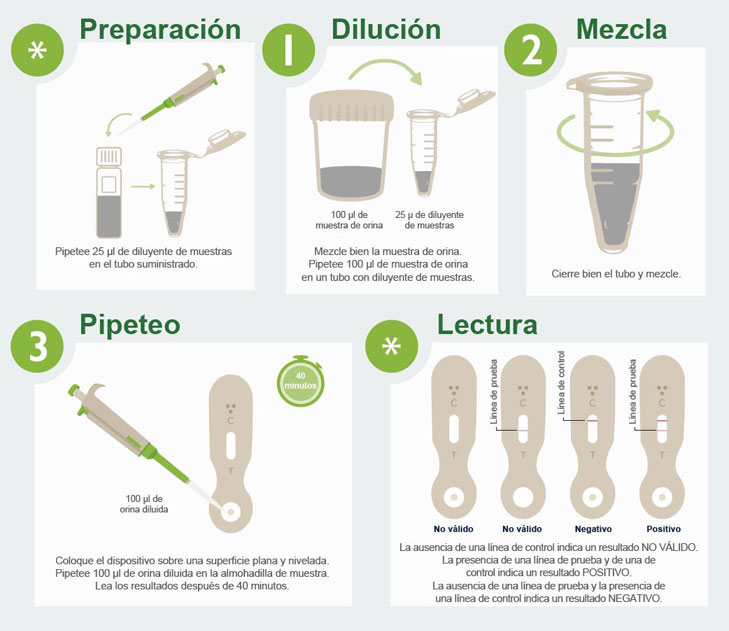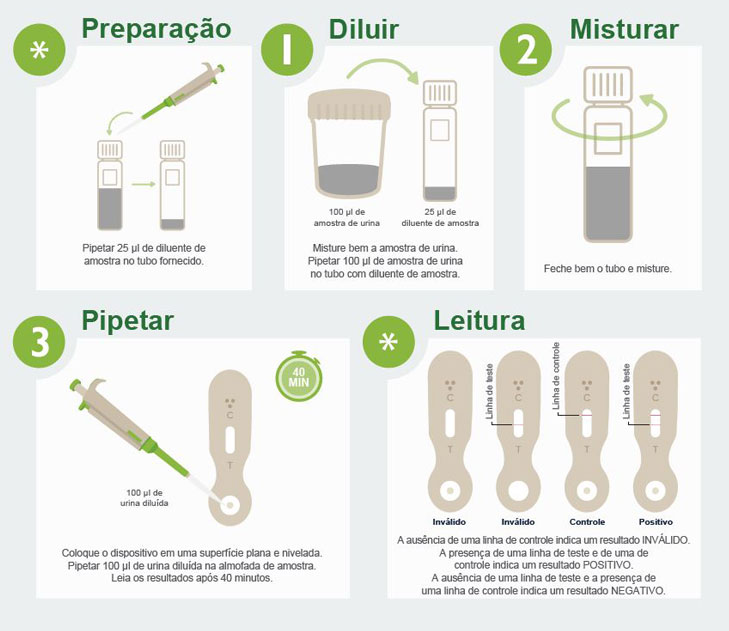
IVD Devices: Histoplasma Urine Antigen LFA
MiraVista Diagnostics’ dedication to innovation and improved accessibility to high quality fungal diagnostics has created a CE marked Histoplasma Urine Antigen LFA.
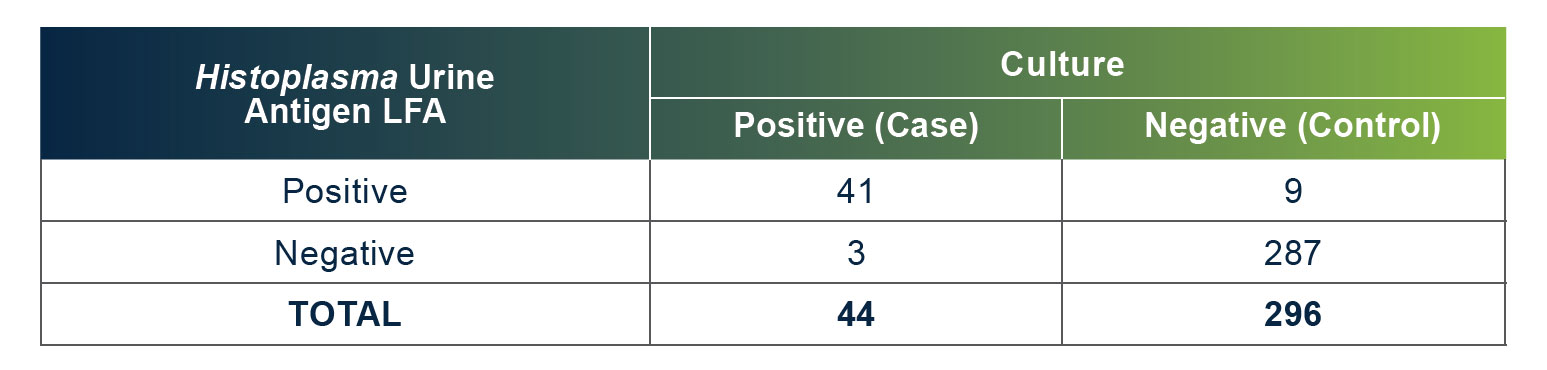
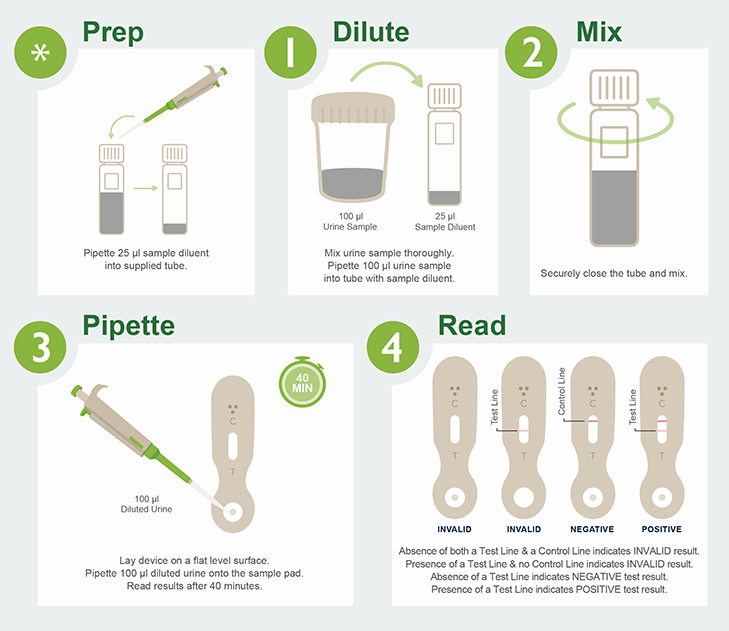
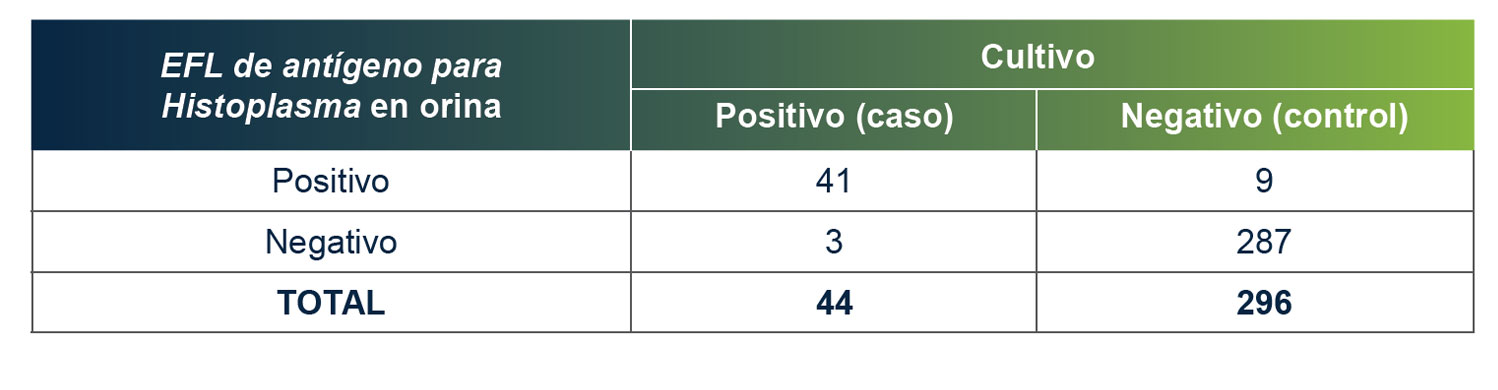
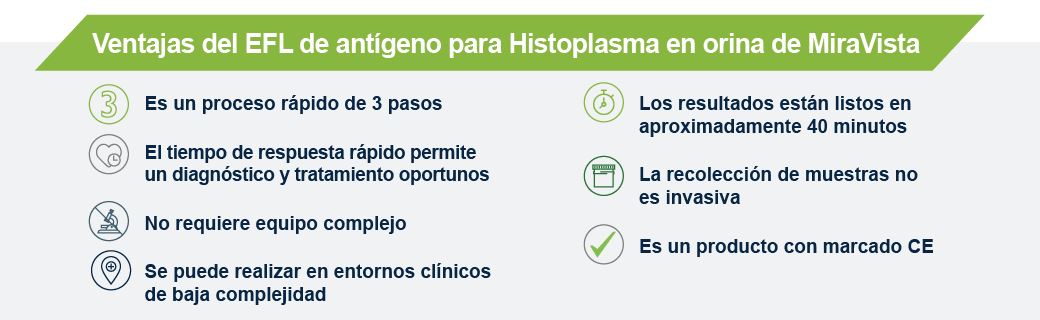
MiraVista Diagnostics is leading the industry with a new Histoplasma Urine Antigen LFA. The rapid, non-invasive test has a proven clinical sensitivity of 93.18% and 96.97% specificity relative to culture proven cases, can be operated in low complexity clinical settings and requires no additional equipment.
Dispositivos de DIV: EFL de antígeno para Histoplasma en orina
La dedicación de MiraVista Diagnostics a la innovación y la accesibilidad mejorada para realizar diagnósticos fúngicos de alta calidad produjo un EFL de antígeno para Histoplasma en orina con marcado CE.
MiraVista Diagnostics lidera la industria con un nuevo EFL de antígeno para Histoplasma en orina. La prueba rápida y no invasiva tiene una sensibilidad clínica comprobada del 93,18 % y un 96,97 % de especificidad en relación con los casos comprobados por cultivo, se puede operar en entornos clínicos de baja complejidad y no requiere equipo de laboratorio adicional.
Dispositivos de DIV: EFL de antígeno urinário para Histoplasma
A dedicação da MiraVista Diagnostics à inovação e melhor acessibilidade a diagnósticos de fungos de alta qualidade criou um EFL de antígeno urinário para Histoplasma com marcação CE.
A MiraVista Diagnostics é líder do setor com o novo EFL de antígeno urinário para Histoplasma. O teste rápido e não invasivo tem sensibilidade clínica comprovada de 93,18% e especificidade de 96,97% em relação aos casos comprovados por cultura, pode ser realizado em ambientes clínicos de baixa complexidade e não requer equipamentos adicionais.